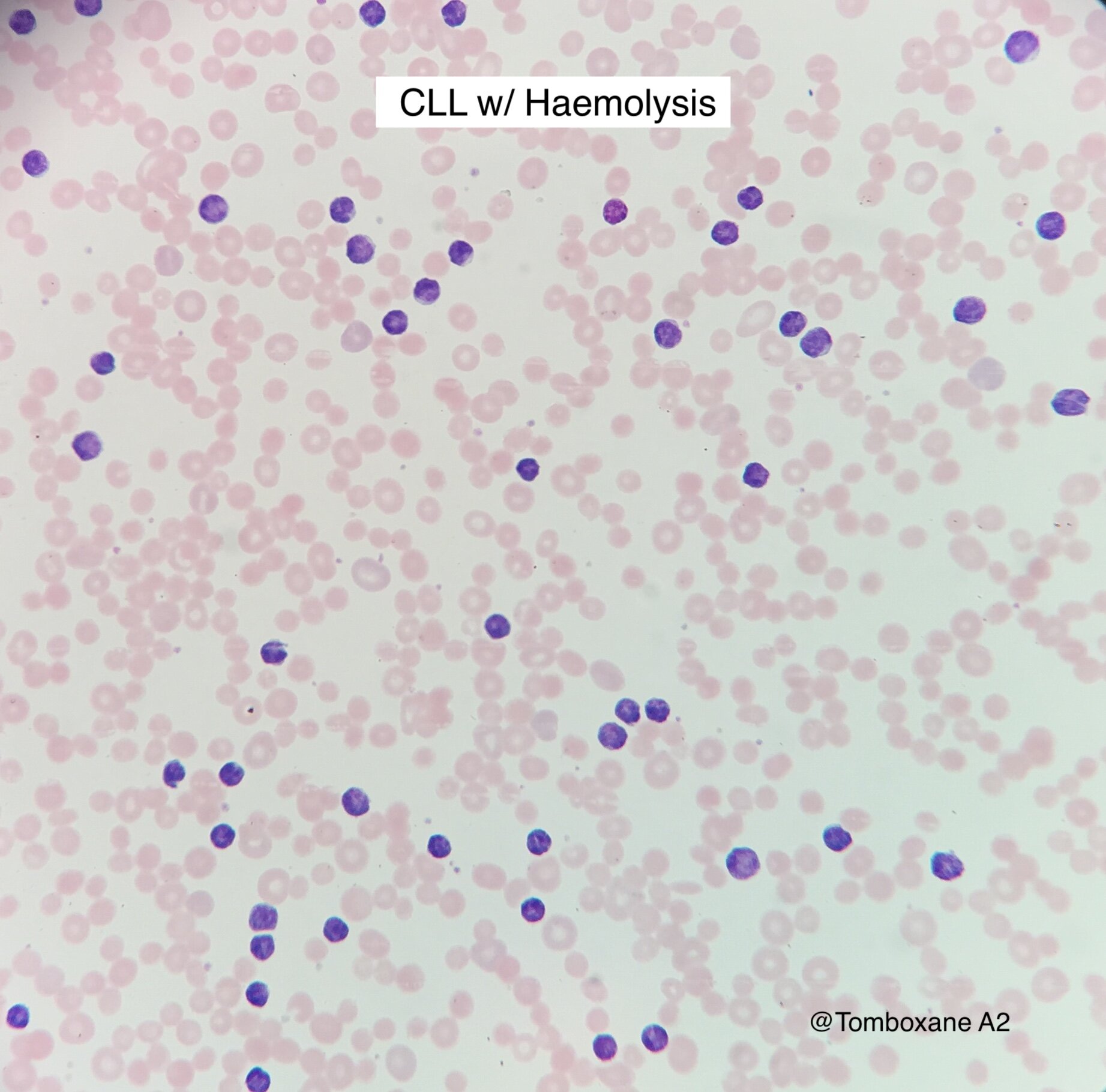
Intro
Incidence 4.2 per 100,000 per year in UK
Median age of presentation 72
Men > Women
Caucasian > other ethnic groups
>80% incidental diagnosis
Diagnosis
Diagnostic criteria
CLL = >5 x10e9/l circulating clonal B cells persisting for >3 months and of characteristic immunophenotype.
CLL Score (now outdated for many centres, as different CD markers often used)
Surface Ig weak, CD5+, CD23+, CD79b-, FMC7-
One point for each
92% of CLL scores 4 or 5
DDx for CD5+ LPD with a low CLL score
atypical CLL, mantle cell, marginal zone
Prolymphocytes:
>15% prolymphocytes = Prolymphocytic progression of CLL (as of 2022, this replaces B-PLL)
cMBL = clinical CD5+ monoclonal B cell lymphocytosis
MBL detectable on a routine FBC but <5 x109/l monoclonal cells
WHO 2016 splits <0.5x109/l (no concern) from 0.5-5 x109/l (annual FBC monitoring)
Staging
BINET
A: <3 lymphoid areas
B: 3 or more lymphoid areas
C: Hb <100 or Plt <100
(Five lymphoid areas are: uni or bilat. cervical, axillary, inguinal, hepatomeg, splenomeg)
RAI
0: Low risk: Lymphocytosis only
I: Intermediate: Lymphadenopathy
II: Intermediate: Hepatomeg or splenomeg + lymphocytosis
III/IV: High risk: Hb <110 or Plt <100
Investigations
Asymptomatic Stage A patients
FBC, reticulocyte, DAT, biochemistry, immunoglobulins, Hep B/C, HIV
Flow cytometry
TP53 deletion (FISH)
Lymph node biopsy only if diagnostic uncertainty
CT not required at diagnosis (perform later pre-treatment)
Diagnosing transformation
5-15% of CLL transforms to DLBCL-like or HL-like
May be localized, biopsy the suspicious site
PET-CT may help identify best biopsy site.
Assessing prognosis
Binet / Rai systems predict outcome in advanced stage patients, but poor for early stage
Other factors:
Age, Gender, PS, co-morbidities
Marrow failure, immunodeficiency, autoimmunity, biomarkers
Type of treatment, Response, Toxicity, MRD status
Prognostic data should not influence timing of first treatment
Biomarkers include IGHV gene analysis, B2M, CD38 positivity
TP53
TP53 loss (del 17p13.1) occurs in 5-10% of patients at diagnosis
TP53 mutation occurs in an additional 5% at diagnosis, 12% of refractory patients
Both TP53 loss and mutation indicate high risk disease, associated with shorter PFS when treated with fixed duration therapies
IGHV
Mutated IGHV associated with long remissions with all therapies, but particularly Ven-O and Ven+I
Unmutated IGHV associated with earlier disease progression in patients receiving fixed duration therapy
IGHV status appears to have less effect on outcomes in continuous therapy
IGHV mutational status is fixed at time of malignant transformation and so testing does not need to be repeated at relapse.
Management
Active Monitoring for Asymptomatic / Early-Stage Disease
6 monthly review in first year to assess rate of progress + address patient and family concerns around leukaemia diagnosis and implications. Then annually if no concern, with GP or hospital
Offer ‘Prehabilitation’ w/ healthy eating, exercise, emotional well-being, optimising co-morbidities
Vaccination - Seasonal ‘Flu, COVID boosters, Pneumococcal, Shingles
CLL12 Trial 2022 - Ibrutinib vs Active Monitoring for asymptomatic early-stage CLL - no difference in 5-yr overall survival (93% in both groups). Higher rate of adverse toxicity in ibrutinib arm.
Indications to treat:
Lymphocyte doubling time <6 months (provided starting Lymph count >30)
Lymphocyte count increased >50% in 2 months
Progressive marrow failure (e.g. Hb <100, Plt <100)
Massive, or symptomatic, splenomegaly (>6cm below costal margin)
Massive, or symptomatic, lymph nodes (>10cm)
Autoimmune anaemia / thrombocytopenia not controlled by standard therapy
Constitutional symptoms
Wt loss >10% in 6 months, fatigue (PS 2 or worse), Fever >38 for 2 wks, night sweats for >1 month.
(NOT indications to treat - High lymphocyte count (in absence of rapid doubling time), hyperviscosity and hypogammaglobulinaemia (in absence of recurrent infection))
Choosing therapy
No single choice, patient-by-patient
Consider CLL biology, co-morbidities / medications, patient choice
Chemoimmunotherapy is no longer indicated when other treatment options are available
Important to achieve maximal response
Achieving MRD negative remission is an independent marker for OS and PFS
Front-line Treament Options (Subject to frequent change!)
Targeted therapies +/- Immunotherapy have replaced traditional cytotoxic chemotherapy and have softened the distinctions between fit / unfit and TP53/noTP53. However some NICE approvals are still based on these groups.
UK Patients without TP53 disruption
Venetoclax-Ibrutinib, fixed duration (15 months) (NICE 2023, CAPTIVATE 2022, GLOW 2022)
Younger, fit patients (CAPTIVATE) = 5-yr OS 96%, 5-yr PF 67%, uMRD 77% at 3 months
Older, unfit patients (GLOW) = 4.5yr PFS 66%. 50% of unmutated IGHV relapsed within 3yrs
Venetoclax-Obinutuzumab, fixed duration (12 months) (NICE 2020, CLL13, CLL14 + f/up 1 yr later)
Younger, fit patients (CLL13) = 3-yr PFS 87%
Older, unfit patients (CLL14) = 6-yr OS 78%
BTKI inhibitors, continuous
Zanubrutinib (NICE 2023, SEQUOIA 2022). 3.5-yr PFS 82% (TP53 pts excluded)
Acalabrutinib (NICE 2021, ELEVATE-TN). 6-yr PFS 62%.
UK Patients with 17p / TP53 disruption (deletions and/or mutations)
BTKI inhibitors, continuous
Current evidence suggests continuous therapy is preferable vs fixed duration
Zanubrutinib (NICE 2023). 3.5-yr PFS 79% for TP53 patients
Acalabrutinib (NICE 2021, ELEVATE-TN). 6-yr PFS 56% for TP53 cohort
Venetoclax-Ibrutinib, fixed duration (15 months) (NICE 2023, CAPTIVATE 2022, GLOW 2022)
4.5yr PFS 45% for TP53 patients in CAPTIVATE
Venetoclax-Obinutuzumab, fixed duration (12 months) (NICE 2020, CLL14 + f/up 1 yr later)
median PFS 4.7 years in TP53 patients
Venetoclax, continuous, approved only if BTKi is unsuitable (NICE 2022)
Retrospective, small studies. 4-yr PFS 84% in one small study
Other approved options, less commonly used first line in UK now:
R-Idelalisib - acceptable alternative if specific issues prevent others (e.g. cardiac disease, warfarin use)
Obinutuzumab-Chlorambucil (CLL11 - PFS and TTNT benefit vs R-Chlormabucil)
FCR (CLL10)
R-Bendamustine
Not yet approved in UK / Emerging therapies
Not currently approved in UK as of Oct 2025 - Ven-Acala, Ven-Acala-Obinu (AMPLIFY 2024)
MRD-guided length of V-I potential promising option for the future, under investigation
Re-treatment with fixed duration regimens for TP53 pts with relapse who received fixed duration first line, under investigation
Relapse/Refractory Options (Subject to frequent change!)
Choosing Therapy
Re-test TP53 at relapse
Consider BTK and PLCG2 mutation testing for patients relapsing whilst on BTKi
Consider time since last treatment and whether relapse occurred on or off treatment, as re-treatment can be effective in patients who have completed a fixed dose regimen and then relapse once off treatment.
Options (Guideline has recommendations on sequencing):
BTK inhibitors, continuous
Zanubrutinib (NICE 2023, ALPINE 2023). 3-yr PFS 65%.
Acalabrutinib (ASCEND, ELEVATE-RR, NICE 2021). Median PFS 3yrs in ELEVATE-RR
Venetoclax-Rituximab, fixed duration (2yrs) (MURANO 2018, NICE 2019)
Wide variation in outcomes - 5-yr PFS 28% vs 72% for unmutated vs mutated IGHV, and 27% vs 42% for disrupted vs wild type TP53
Only 1% of patients in MURANO received a BTKi as first line treatment.
Venetoclax monotherapy, continuous
Rituximab-Idelasilib
Not yet approved in UK / Emerging therapies
Pirtobrutinib (BRUIN321 2024). Median PFS of 15months in heavily pre-treated patients
CAR-T therapy under investigation
When to consider allogeneic stem cell transplant?
Complex, individualised decision. ??Fit patients with high risk disease and multiple treatment failures
Supportive Care
Anti-microbial prophylaxis
Consider PCP prophylaxis for at least first 12 months of BTKi
Consider Aciclovir prophylaxis for patients with a history of VZV/HSV1 infections
Azoles avoided in combination with BTKi and BCL2i due to interactions
Hypogammagloublinaemia
Prophylactic antibiotics for recurrent infections
Consider immunoglobulin replacement 3-4 weekly if low IgG (<4g/l) and recurrent infection. Discontinue after 1 year if not reduction in rates of infection.
Vaccination
Annual flu vaccination + COVID-19 boosters
Pneumococcal vaccination - Prevnar (PCV) 13 or 15, followed by Pneumovax23 two months later (watch this space: PCV20 may replace the above in 2026).
Shingles - non-live vaccine now available (Shingrix)
Drugs: A few notes
Covalent Bruton Tyrosine Kinase inhibitors (BTKi) (Also see here)
Ibrutinib, Alcalabrutinib, Zanubrutinib
SE: acquired platelet function defect, hold for 5-7 days before interventional procedures / surgery
SE: Petechial rash, easy bruising & arthralgia usually improve with time
SE: Hypertension, AF & VT (including sudden cardiac death)
SE: With alcalabrutinib also look for headache, gastritis. But probabaly less cardiac side effects than ibrutinib (N.B. new tablet formulation of acala no longer has an issue with concurrent use of PPI’s)
Avoid CYP3A4 inhibitors - warfarin, fish oils, Vitamin E, St Johns Wort
Grapefruit and Seville oranges must be avoided
Non-Covalent BTKi
e.g. Pirtobrutinib
Alternative, reversible mechanism of binding BTK compared to covalent inhibitors (review paper 2021)
—> Can still be effective after resistance has developed to covalent BTKi’s
Typical BTKi side effects (bruising, rash, arthralgai, AF) may be less common and less severe.
BCL2 inhibtior - Venetoclax
MOA: Inhibitor of BCL2. BCL2 is a anti-apoptotic protein that is overexpressed in cells of many haematological malignancies. Apoptosis is reduced in these cells because BCL2 sequesters the pro-apoptotic protein BAX. —> Inihibiting BCL2 releases BAX which leads to cell death.
Significant risk of tumour lysis (—> dose escalated slowly over course of first cycle)
Ibrutinib + Venetoclax
Proposed to be synergistic when given in combination
Ibrutinib drives CLL out of lymph nodes, into peripheral blood where more sensitive to venetoclax
BTK inhibition makes CLL cells more dependent on BCL2 —> increased sensitivity to venetoclax
A 3 month lead in with ibrutinib reduces the risk of tumour lysis syndrome associated with venetoclax
PI3 Kinase Inhibitors - Idelasilib
May trigger life-threatening pneumonitis or colitis
Monitor liver function
Monitor CMV PCR’s
Use PCP prophylaxis
A Few Trial notes
Phase 3, First line CLL for fit patients without TP53 disruption
926 patiens. 1:1:1:1 randomisation:
FCRx6 or R-Benda x6
R-Venetoclax x12
O-Venetoclax x12
Venetolax-O-Ibrutinib x12
At 15 months, uMRD V-O-I (92%) & O-V (86%) vs R-V (57%) & Chemo (53%)
3-yr PFS was V-O-I (90.5%) & O-V (87.7%) vs (80%) & Chemo (75.5%)
Grade 3/4 infection was V-O-I (21%) vs O-V (13%)
Phase 3, R/R CLL, Ibrutinib vs Zanubritinib
2yr PFS: 78% zanubrutinib, 65% ibrutinib
Rates of drug discontinuation + cardiac events lower with zanubrutinib vs ibrutinib
Phase 3, First line CLL. Ibrutinib+Venetoclax vs O-Chlormabucil
211 patient, >65 yo (or high frailty score (CIRS) or CrCl <70ml/min)
Excluded del17p and TP53 mutation patients.
undetectable MRD (uMRD) I+V 55% in BM
2-yr I+V OS 90%, PFS 84%
Phase 3, First line CLL. Zanubrutinib vs R-Benda except all 17p del got Zanu (arm C)
590 patients, >65yo or <65 with co-morbidity
At 2 yrs, median PFS not reached for either arm but superior for zanubrutinib
50% neutropenia with zanubrutinib
Phase 2, First line CLL. 15 months Ibrutinib + 12 months Venetoclax
160 patients, <70 yo only in trial. 92% completed full treatment.
CR 55%. For those in CR, undetectable MRD (uMRD) was 90% in PB and 72% in BM.
2-yr OS 98%, PFS 95%
Phase 3, First Line CLL. O-Acala vs Acala va O-Chlorambucil
535 patients, >65 yo
2yr PFS 93% O-A, 87% A, 47% O-C
5 yr f/up: Median OS not yet reached for any arm. Est. 60 month OS 90% A-O, 84% A, 82% O-C
Phase 2, First line CLL. Up to 24 cycles of ibrutinib + venetoclax
80 high risk patients (del17p, TP53 mutation, del11q, unmutated IGHV or Age >65)
CR 88%. Undetectable MRD (uMRD) was 61% in BM
Only 3 patients completed 24 cycles of treatment.
CLL14 2019
12 months Venetoclax + 6x Obinutuzumab vs. 12 months Chlorambucil + 6x Obinutuzumab
432 patients
O-Venetoclax showed higher rates of complete response, MRD negativity and longer PFS
Follow-up off treatment confirmed longer PFS (median PFS not reached in O-V, 35 months for O-C)
Phase 3, R/R CLL. R-Venetoclax vs R-Benda
389 patients
2-yr PFS 84.9% R-Ven vs 36.3% R-Benda
COMPLEMENT-1 2015
Chlorambucil-Ofatunumab vs Chlormabucil alone
Combo better PFS but no difference in OS
Hard to compare to CLL11 as different dose of chlorambucil used
CLL11 2014
Chlorambucil-Obinutuzumab vs Chlorambucil-Rituximab vs Chlormabucil alone
Chlorambucil-Obinutuzumab had superior PFS and TTNT
Infusion reactions more common with obinutuzumab
CLL10 2013
FCR x6 vs R-Benda x6
FCR superior for ORR, MRD-neg remissions, length of first remission in young, fit patients
FCR arm had more serious adverse events
Overall survival similar for both arms